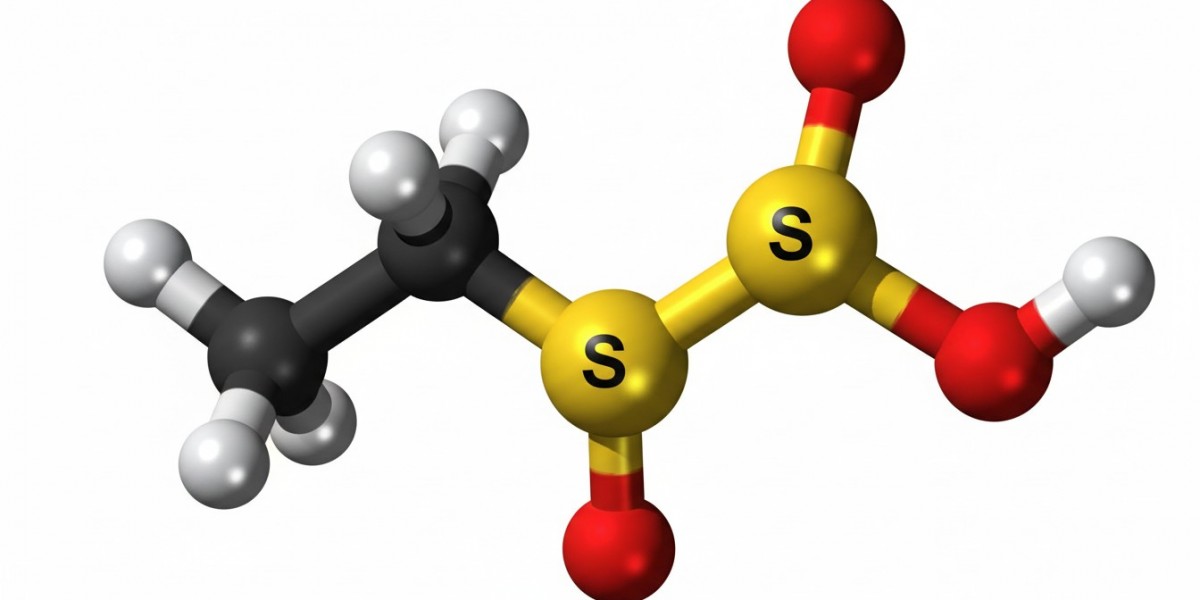
Ethylene sulfate is an organic chemical compound that has gained attention due to its unique structure and functional properties. It belongs to a class of compounds known as sulfate esters, which are formed by the reaction of alcohols with sulfuric acid. This compound appears as a colorless to pale yellow liquid and is primarily used in research and industrial processes. Its chemical behavior makes it valuable in synthesis and various chemical transformations, particularly where reactivity and stability must be carefully balanced.
Chemical Structure and Composition
The molecular structure of ethylene sulfate consists of an ethylene group bonded to a sulfate functional group. This arrangement gives the compound a cyclic structure, which contributes to its chemical stability compared to other sulfate esters. The presence of oxygen and sulfur atoms creates a polar molecule, allowing it to participate in a wide range of chemical reactions. Its structural uniqueness also makes it useful as an intermediate in the preparation of other organic compounds.
Physical Properties
Ethylene sulfate typically exists as a clear liquid under standard conditions. It has a relatively high boiling point due to strong intermolecular forces, particularly dipole interactions. The compound is moderately soluble in water and highly soluble in many organic solvents. Its density is slightly higher than water, and it does not have a strong odor. These physical properties make it suitable for controlled laboratory applications where stability and consistency are important.
Chemical Behavior
One of the key characteristics of ethylene sulfate is its reactivity. It can undergo hydrolysis when exposed to water, breaking down into ethylene glycol and sulfuric acid derivatives. This reaction is important in understanding its storage and handling requirements. Additionally, it can participate in substitution reactions, making it a useful reagent in organic synthesis. Its cyclic sulfate structure allows it to act as an electrophile, reacting with nucleophiles in various chemical processes.
Methods of Preparation
Ethylene sulfate is typically synthesized through the reaction of ethylene glycol with sulfuryl chloride or sulfuric acid derivatives. The process involves careful control of temperature and reaction conditions to ensure the formation of the desired cyclic structure. Another method involves the oxidation of ethylene sulfite, which produces ethylene sulfate as a product. These preparation methods are mainly carried out in controlled industrial or laboratory environments.
Industrial Applications
In industrial settings, ethylene sulfate is used as an intermediate in the production of specialty chemicals. It plays a role in the synthesis of polymers, surfactants, and other functional materials. Its ability to react with different chemical agents makes it valuable in manufacturing processes that require precise chemical transformations. Additionally, it is sometimes used in the development of coatings and additives that enhance material performance.
Laboratory Uses
In research laboratories, ethylene sulfate is used as a reagent for studying reaction mechanisms and developing new synthetic pathways. Chemists often use it to introduce sulfate groups into organic molecules or to test the reactivity of different compounds. Its predictable behavior under controlled conditions makes it a reliable tool for experimental work. It is also used in academic research to explore the properties of cyclic sulfate compounds.
Safety and Handling
Handling ethylene sulfate requires proper safety precautions. The compound can be harmful if inhaled, ingested, or absorbed through the skin. It may cause irritation to the eyes, skin, and respiratory system. Therefore, it should be handled in a well-ventilated area, preferably under a fume hood. Protective equipment such as gloves, goggles, and lab coats should always be used. Proper storage in sealed containers is also essential to prevent contamination and degradation.
Environmental Impact
The environmental impact of ethylene sulfate depends on how it is used and disposed of. If released into the environment, it can break down into other chemical substances, some of which may affect soil and water quality. Proper waste management practices are necessary to minimize its impact. Industries that use this compound are required to follow environmental regulations to ensure safe disposal and prevent pollution.
Advantages and Limitations
Ethylene sulfate offers several advantages, including its stability, reactivity, and versatility in chemical synthesis. It can be used in a wide range of applications, making it a valuable compound in both industrial and laboratory settings. However, it also has limitations, such as potential health risks and sensitivity to moisture. These factors must be considered when working with the compound to ensure safe and effective use.
Future Prospects
The future of ethylene sulfate lies in its potential applications in advanced materials and green chemistry. Researchers are exploring ways to use it in more sustainable chemical processes, reducing environmental impact while maintaining efficiency. Its role in developing new polymers and functional materials is also an area of ongoing research. As technology advances, the demand for such versatile compounds is likely to increase.
Conclusion
Ethylene sulfate is an important chemical compound with a wide range of applications in industry and research. Its unique structure and properties make it a valuable tool for chemists and manufacturers alike. While it offers many benefits, it must be handled with care due to its potential hazards. With proper use and continued research, ethylene sulfate will remain a significant component in the field of chemistry.